Background: Hematopoietic cell transplantation (HCT) is an integral part of the treatment of multiple myeloma (MM). While autologous stem cell transplantation (auto-HCT) is most commonly used, the duration of response is typically finite. Allogeneic HCT (allo-HCT) can provide prolonged survival in some patients, given the added benefit of the graft-versus-myeloma effect. However, long-term data is needed to show this improvement.
Method: We retrospectively reviewed a cohort of 37 consecutive patients with newly diagnosed MM who received allo-HCT as part of consolidation therapy between 1994 to 2016.
Results: The median age was 54 years (range, 32 to 68), and 54% were male. The Revised International Staging System (R-ISS) stages were I, II, III, and unknown in 27%, 38%, 11%, and 24% of patients, respectively. High-risk cytogenetics (IMWG definition) was identified in 22% of patients. The median time from diagnosis to allo-HCT was 8.8 months (range; 3.3 to 34.3). For induction treatment, fourteen patients (38%) received a combination of immunomodulatory drug (IMiD) plus proteasome inhibitor (PI), sixteen patients (43%) received either IMiD or PI in combination with other agents, and seven patients (19%) did not receive either an IMiD or PI. Twenty-seven (73%) patients received auto-HCT before allo-HCT. Thirty-four (92%) patients received allo-HCT as part of various clinical trials. Median time from auto-HCT to allo-HCT was 4 months (2.5 to 27.3). Prior to allo-HCT, 1 (3%) patient was in complete remission (CR), 18 (48.5%) were in very good partial remission (VGPR), and 18 (48.5%) were in partial remission (PR). Twenty-three (62%) patients received non-myeloablative (NMA) conditioning, 10 (27%) reduced-intensity (RIC), and 4 (11%) myeloablative conditioning (MAC). The graft source was matched unrelated (MUD) in 16% and matched sibling donor (MRD) in 84% of patients. Ten (27%) patients received maintenance therapy after allo-HCT, including bortezomib (n=2), thalidomide (n=2), ixazomib (n=2), and lenalidomide (n=4).
The median days to neutrophil and platelet engraftment was 12 (ANC ≥500/µL_ range; 10 to 59) and 13 (platelet count ≥20K/µL _range; 9 to 70), respectively. The cumulative incidence (CI) of non-relapse mortality (NRM) was 16% at 1-year and 19% at 3-years after allo-HCT. There was no difference in NRM between MAC or NMA/RIC conditioning. The overall response rate (PR or better) was 97%, with a 54% stringent CR+CR rate. The incidence of grade I-IV acute graft-versus-host disease (GVHD) was 35%, while chronic GVHD was seen in 62%. Causes of death were deemed to be disease-related in 8 patients, treatment-related in 11 patients, and 1 unknown.
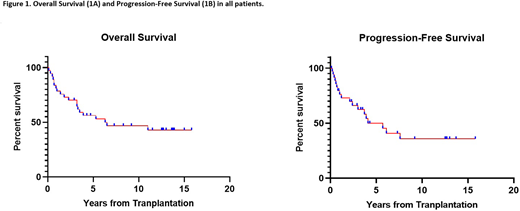
The median follow-up in surviving patients was 12.6 years (range; 2.8 to 15.8 years). The 3, 5, and 10-year actuarial overall survival (OS) rates were 70%, 56%, and 47%, respectively (Figure 1A). The 3, 5, and 10-year actuarial progression-free survival (PFS) rates were 66%, 50%, and 36%, respectively (Figure 1B). At the last follow up, 46% (n=17) of patients were alive in the entire cohort, 65% (n=11) of which survived for longer than 10-years from transplant. Sixteen percent (n=6) remained alive and in continued remission for more than 10 years from transplant, one-third of whom received maintenance treatment post allo-HCT. The longest ongoing remission was 15.8 years in this cohort.
Conclusion: Allo-HCT may result in durable (>10 years) remission in a number of MM patients when performed early in the disease course. Larger studies would help identify the patients who would benefit the most, given the risk of graft-versus-host disease after allo-HCT.
Popat:Bayer: Research Funding; Novartis: Research Funding. Kebriaei:Pfizer: Other: Served on advisory board; Kite: Other: Served on advisory board; Amgen: Other: Research Support; Jazz: Consultancy; Novartis: Other: Served on advisory board; Ziopharm: Other: Research Support. Oran:Celgene: Consultancy; ASTEX: Research Funding; Arog Pharmaceuticals: Research Funding. Hosing:NKARTA Inc.: Consultancy. Manasanch:Adaptive Biotechnologies: Honoraria; GSK: Honoraria; Sanofi: Honoraria; BMS: Honoraria; Takeda: Honoraria; Quest Diagnostics: Research Funding; Merck: Research Funding; JW Pharma: Research Funding; Novartis: Research Funding; Sanofi: Research Funding. Lee:Amgen: Consultancy, Research Funding; Genentech: Consultancy; Regeneron: Research Funding; Daiichi Sankyo: Research Funding; Sanofi: Consultancy; GlaxoSmithKline: Consultancy, Research Funding; Genentech: Consultancy; Takeda: Consultancy, Research Funding; Janssen: Consultancy, Research Funding; Celgene: Consultancy, Research Funding. Kaufman:Karyopharm: Honoraria; Bristol Myers Squibb: Research Funding; Janssen: Research Funding. Patel:Precision Biosciences: Research Funding; Takeda: Consultancy, Research Funding; Cellectis: Research Funding; Janssen: Consultancy, Research Funding; Poseida: Research Funding; Oncopeptides: Consultancy; Nektar: Consultancy, Research Funding; Celgene: Consultancy, Research Funding; Bristol Myers Squibb: Consultancy, Research Funding. Orlowski:Founder of Asylia Therapeutics, Inc., with associated patents and an equity interest, though this technology does not bear on the current submission.: Current equity holder in private company, Patents & Royalties; STATinMED Research: Consultancy; Sanofi-Aventis, Servier, Takeda Pharmaceuticals North America, Inc.: Honoraria, Membership on an entity's Board of Directors or advisory committees; Laboratory research funding from BioTheryX, and clinical research funding from CARsgen Therapeutics, Celgene, Exelixis, Janssen Biotech, Sanofi-Aventis, Takeda Pharmaceuticals North America, Inc.: Research Funding; Amgen, Inc., AstraZeneca, BMS, Celgene, EcoR1 Capital LLC, Forma Therapeutics, Genzyme, GSK Biologicals, Ionis Pharmaceuticals, Inc., Janssen Biotech, Juno Therapeutics, Kite Pharma, Legend Biotech USA, Molecular Partners, Regeneron Pharmaceuticals, Inc.,: Honoraria, Membership on an entity's Board of Directors or advisory committees. Thomas:Ascentage: Membership on an entity's Board of Directors or advisory committees, Research Funding; BMS: Research Funding; X4 Pharma: Research Funding; Pharmacyclics: Other: Advisory Boards; Xencor: Research Funding; Genentech: Research Funding. Shpall:Takeda: Other: Licensing Agreement; Magenta: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees; Celgene: Membership on an entity's Board of Directors or advisory committees; Adaptimmune: Membership on an entity's Board of Directors or advisory committees; Zelluna: Membership on an entity's Board of Directors or advisory committees. Champlin:Takeda: Patents & Royalties; Actinium: Consultancy; Johnson and Johnson: Consultancy; Omeros: Consultancy; Cytonus: Consultancy; DKMS America: Membership on an entity's Board of Directors or advisory committees; Genzyme: Speakers Bureau. Qazilbash:Bioclinica: Consultancy; Amgen: Research Funding; Angiocrine: Research Funding; Bioline: Research Funding; Janssen: Research Funding. Bashir:Celgene: Research Funding; Amgen: Other: Advisory Board; Purdue: Other: Advisory Board; Takeda: Other: Advisory Board, Research Funding; Acrotech: Research Funding; StemLine: Research Funding; KITE: Other: Advisory Board.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal